
It is estimated that about 30% of soluble cytosolic proteins contain this CMA-targeting motif. Only proteins bearing a particular targeting motif in their amino acid sequence, biochemically related to the pentapeptide KFERQ ( 2), are selectively recognized by the heat shock cognate protein of 70 kDa (hsc70), the chaperone that mediates their delivery to lysosomes for their degradation via CMA ( 3). Two properties differentiate chaperone-mediated autophagy (CMA) from the other types of autophagy in mammalian cells: its selectivity towards a particular pool of cytosolic proteins and the mechanism of delivery of the substrate proteins to lysosomes ( 1). However, the most unequivocal method to measure CMA is by directly tracking the translocation of substrate proteins into isolated lysosomes. Changes in the levels/activity of the lysosomal components required for substrate translocation can be used to stimulate CMA activity. 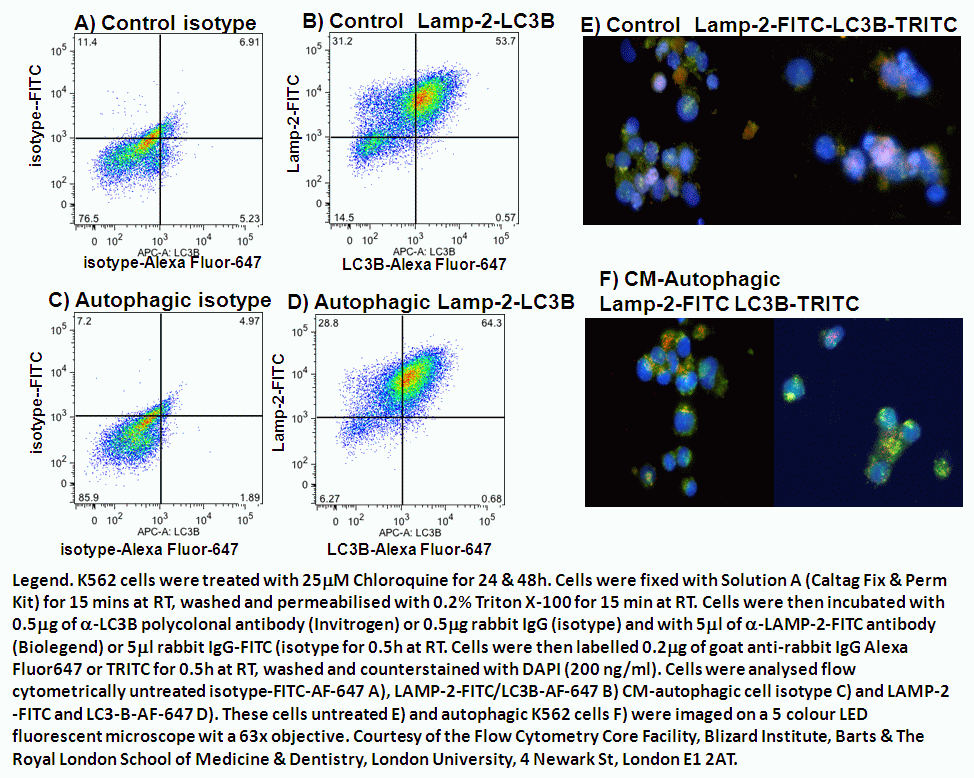 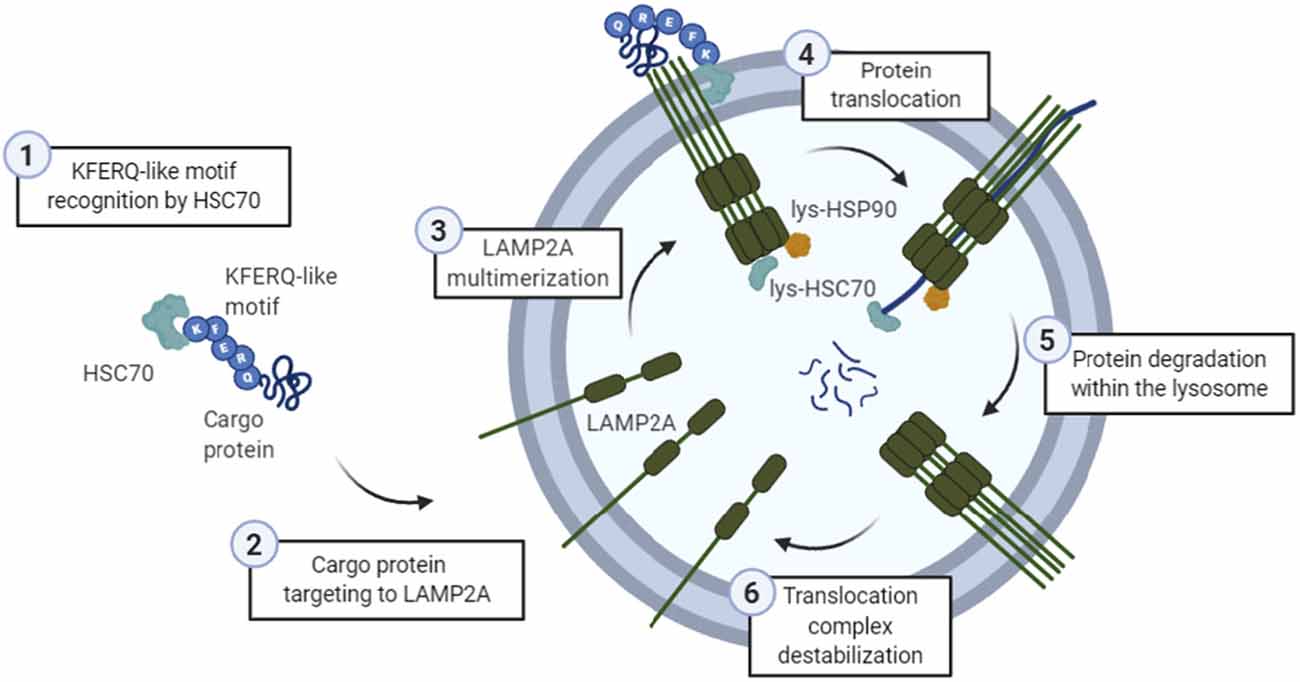
In contrast to other forms of autophagy, where substrates are engulfed or sequestered along with other cytosolic components, CMA substrates are translocated one-by-one across the lysosomal membrane. Impaired CMA under these conditions may be responsible for the accumulation of damaged proteins inside cells and for their higher vulnerability to stressors. We have found that CMA activity decreases in aging and in some age-related disorders such as Parkinson’s disease. CMA is maximally activated in response to stressors such as prolonged starvation, exposure to toxic compounds, or oxidative stress. Chaperone-mediated autophagy (CMA) is the only type of autophagy in mammalian cells able to selectively degrade cytosolic proteins in lysosomes.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
